Menu
Post-Translational Modifications
This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What are Post-translational Modifications?
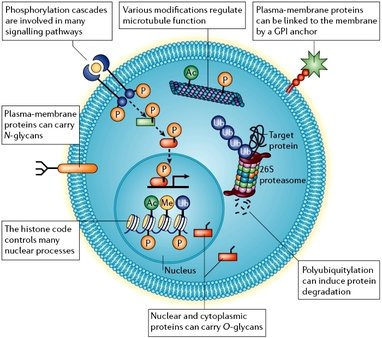
Post-translational modifications involve the addition of a variety of different functional groups to proteins after they are synthesized within the cell (1). These groups can exert many different possible effects, including marking a protein for degradation or trafficking a protein to a particular part of the cell(2,3). Changes in the post-translational modifications of proteins can be monitored using mass spectrometry, which examines mass differences between fragmented peptide chains of proteins in order to identify them (4).
Phosphorylation of AQP2
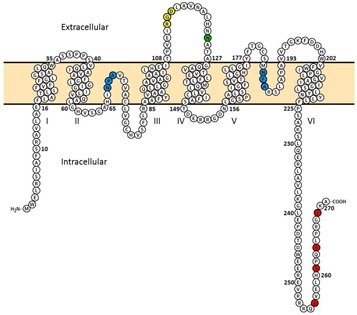
Previous experimental evidence has shown extensive phosphorylation of AQP2 near the protein's C terminus (5). In the figure on the left, the important phosophorylation residues are shown in red. The residue with the most significant effect when phosphorylated is Serine-256(S256). S256 phosophorylation promotes AQP2 movement to the plasma membrane of the collecting duct cell (6). The residues of S269 and S261 have also shown evidence of directing AQP2 localization. S269 when phosphorylated increases the time that AQP2 is within the plasma mebrane, while in contrast S261 when phosphorylated increases the rate at which AQP2 is endocytosed back into the cell (7,8). The function of S264 when phosphorylated is currently poorly understood (5). Bioinformatic analysis using NetPhos confirms these four sites as very likely phosphorylation targets (9).
Ubiquitination of AQP2
Bioinformatic analysis of AQP2 using iPTMnet finds possible ubiquitination sites at lysines 228 and 270 (10). Previous experimental evidence has suggested that ubiquitination on lysine 270 may trigger endocytosis of AQP2 back into the cell from the plasma membrane (11).
Discussion
The wide variety of different sites through which AQP2 expression is controlled gives analysis of post-translational modifications great explanatory power in determining dynamics of AQP2 at the plasma membrane. Analysis of currently uncharacterized sites known to be modified such as S264 provides an opportunity to fine tune the dynamics of the cellular location of AQP2.
Works Cited
1. Post Translational Modifications. http://keck.med.yale.edu/proteomics/technologies/posttransmodifications/. Accessed on April 11th, 2017.
2. Fredrickson, E. K., & Gardner, R. G. (2012). Selective destruction of abnormal proteins by ubiquitin-mediated protein quality control degradation. Seminars in Cell & Developmental Biology, 23(5), 530-537. doi:10.1016/j.semcdb.2011.12.006
3.Reynders, E., Foulquier, F., Annaert, W., & Matthijs, G. (2011). How Golgi glycosylation meets and needs trafficking: the case of the COG complex. Glycobiology, 21(7), 853-863. doi:10.1093/glycob/cwq179
4.Dephoure, N., Gould, K. L., Gygi, S. P., & Kellogg, D. R. (2013). Mapping and analysis of phosphorylation sites: a quick guide for cell biologists. Molecular Biology of the Cell, 24(5), 535-542. doi:10.1091/mbc.E12-09-0677
5.Jung, H. J., & Kwon, T. H. (2016). Molecular mechanisms regulating aquaporin-2 in kidney collecting duct. American Journal of Physiology-Renal Physiology, 311(6), F1318-F1328. doi:10.1152/ajprenal.00485.2016
6.Lu, H. J., Matsuzaki, T., Bouley, R., Hasler, U., Qin, Q. H., & Brown, D. (2008). The phosphorylation state of serine 256 is dominant over that of serine 261 in the regulation of AQP2 trafficking in renal epithelial cells. American Journal of Physiology-Renal Physiology, 295(1), F290-F294. doi:10.1152/ajprenal.00072.2008
7Tamma, G., Robben, J. H., Trimpert, C., Boone, M., & Deen, P. M. T. (2011). Regulation of AQP2 localization by S256 and S261 phosphorylation and ubiquitination. American Journal of Physiology-Cell Physiology, 300(3), C636-C646. doi:10.1152/ajpcell.00433.2009
8.Moeller, H. B., Knepper, M. A., & Fenton, R. A. (2009). Serine 269 phosphorylated aquaporin-2 is targeted to the apical membrane of collecting duct principal cells. Kidney International, 75(3), 295-303. doi:10.1038/ki.2008.505
9. NetPhos 3.1. http://www.cbs.dtu.dk/services/NetPhos/. Accessed on April 11th,2017.
10. iPTMnet. http://research.bioinformatics.udel.edu/iptmnet/. Accessed on April 11th,2017.
11.Moeller, H. B., Aroankins, T. S., Slengerik-Hansen, J., Pisitkun, T., & Fenton, R. A. (2014). Phosphorylation and ubiquitylation are opposing processes that regulate endocytosis of the water channel aquaporin-2. Journal of Cell Science, 127(14), 3174-3183. doi:10.1242/jcs.150680
2. Fredrickson, E. K., & Gardner, R. G. (2012). Selective destruction of abnormal proteins by ubiquitin-mediated protein quality control degradation. Seminars in Cell & Developmental Biology, 23(5), 530-537. doi:10.1016/j.semcdb.2011.12.006
3.Reynders, E., Foulquier, F., Annaert, W., & Matthijs, G. (2011). How Golgi glycosylation meets and needs trafficking: the case of the COG complex. Glycobiology, 21(7), 853-863. doi:10.1093/glycob/cwq179
4.Dephoure, N., Gould, K. L., Gygi, S. P., & Kellogg, D. R. (2013). Mapping and analysis of phosphorylation sites: a quick guide for cell biologists. Molecular Biology of the Cell, 24(5), 535-542. doi:10.1091/mbc.E12-09-0677
5.Jung, H. J., & Kwon, T. H. (2016). Molecular mechanisms regulating aquaporin-2 in kidney collecting duct. American Journal of Physiology-Renal Physiology, 311(6), F1318-F1328. doi:10.1152/ajprenal.00485.2016
6.Lu, H. J., Matsuzaki, T., Bouley, R., Hasler, U., Qin, Q. H., & Brown, D. (2008). The phosphorylation state of serine 256 is dominant over that of serine 261 in the regulation of AQP2 trafficking in renal epithelial cells. American Journal of Physiology-Renal Physiology, 295(1), F290-F294. doi:10.1152/ajprenal.00072.2008
7Tamma, G., Robben, J. H., Trimpert, C., Boone, M., & Deen, P. M. T. (2011). Regulation of AQP2 localization by S256 and S261 phosphorylation and ubiquitination. American Journal of Physiology-Cell Physiology, 300(3), C636-C646. doi:10.1152/ajpcell.00433.2009
8.Moeller, H. B., Knepper, M. A., & Fenton, R. A. (2009). Serine 269 phosphorylated aquaporin-2 is targeted to the apical membrane of collecting duct principal cells. Kidney International, 75(3), 295-303. doi:10.1038/ki.2008.505
9. NetPhos 3.1. http://www.cbs.dtu.dk/services/NetPhos/. Accessed on April 11th,2017.
10. iPTMnet. http://research.bioinformatics.udel.edu/iptmnet/. Accessed on April 11th,2017.
11.Moeller, H. B., Aroankins, T. S., Slengerik-Hansen, J., Pisitkun, T., & Fenton, R. A. (2014). Phosphorylation and ubiquitylation are opposing processes that regulate endocytosis of the water channel aquaporin-2. Journal of Cell Science, 127(14), 3174-3183. doi:10.1242/jcs.150680
